Identification of Cu(100)/Cu(111) Interfaces as Superior Active Sites for CO Dimerization During CO2 Electroreduction | Journal of the American Chemical Society

Identification of Cu(100)/Cu(111) Interfaces as Superior Active Sites for CO Dimerization During CO2 Electroreduction | Journal of the American Chemical Society
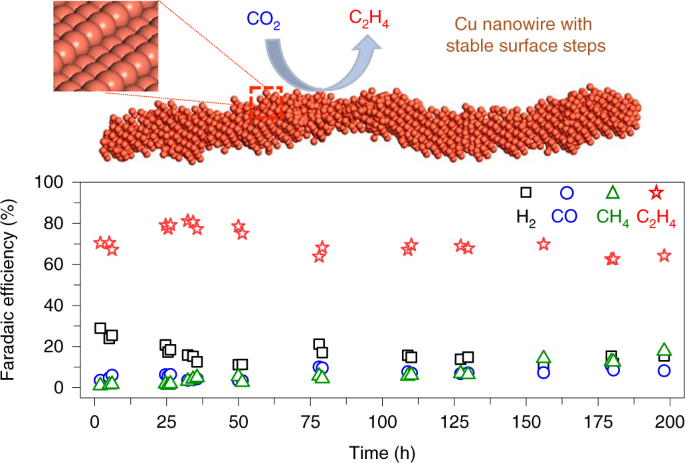
Highly active and stable stepped Cu surface for enhanced electrochemical CO2 reduction to C2H4 | Nature Catalysis

Coupling of Cu(100) and (110) Facets Promotes Carbon Dioxide Conversion to Hydrocarbons and Alcohols - Zhong - 2021 - Angewandte Chemie - Wiley Online Library

Identification of Cu(100)/Cu(111) Interfaces as Superior Active Sites for CO Dimerization During CO2 Electroreduction | Journal of the American Chemical Society

Identification of Cu(100)/Cu(111) Interfaces as Superior Active Sites for CO Dimerization During CO2 Electroreduction | Journal of the American Chemical Society

Kupfer-Elektrode Ø 8 mm x 100 mm, Cu 99,99 | Kupfer | Elemente Aluminium - Zirconium | Polymet - Reine Metalle.
CU Abzweig 72Grad/100/100 CU | Rohr | Formteile Rinne & Rohr | Entwässerung | Dach | Sortiment | Dachdecker-Einkauf Süd eG

Full atomistic reaction mechanism with kinetics for CO reduction on Cu(100) from ab initio molecular dynamics free-energy calculations at 298 K | PNAS

Lithiophilic Faceted Cu(100) Surfaces: High Utilization of Host Surface and Cavities for Lithium Metal Anodes - Gu - 2019 - Angewandte Chemie International Edition - Wiley Online Library