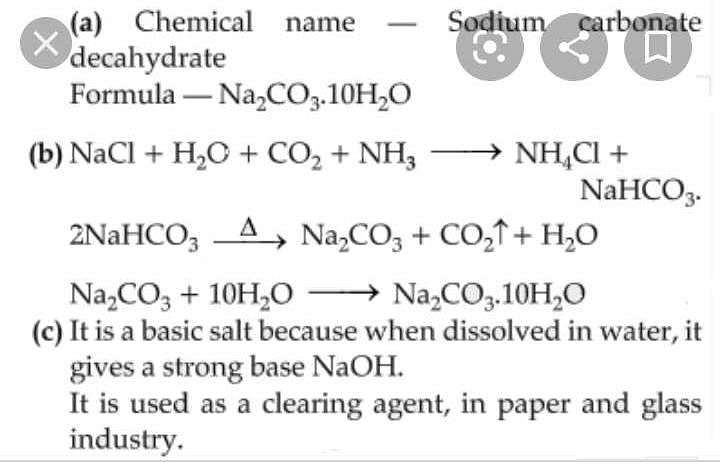
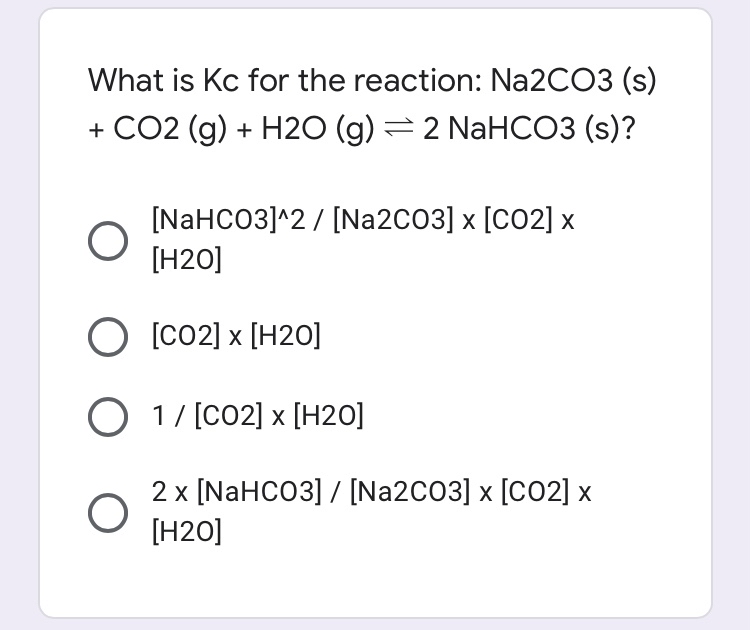![6132-02-1・Sodium Carbonate Decahydrate・191-01545[Detail Information] | [Common Chemicals & Lab Tools]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 6132-02-1・Sodium Carbonate Decahydrate・191-01545[Detail Information] | [Common Chemicals & Lab Tools]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/05/191-01545.png)
6132-02-1・Sodium Carbonate Decahydrate・191-01545[Detail Information] | [Common Chemicals & Lab Tools]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
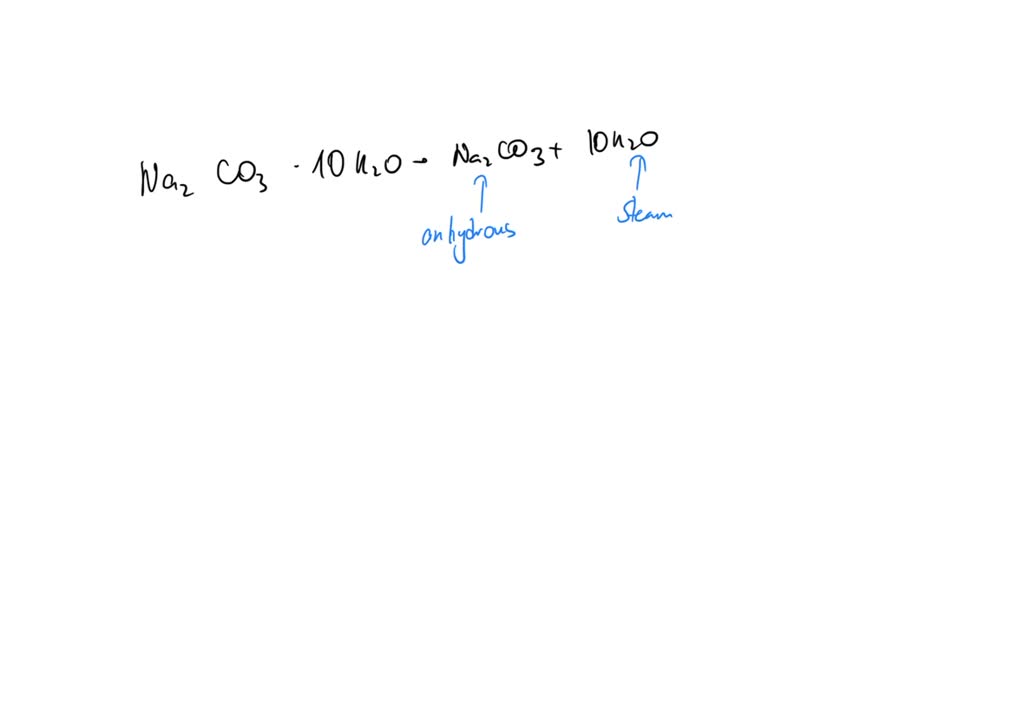
SOLVED: The equation for the effect of heat on hydrated sodium carbonate is as shown. Na2CO3· 10H2O(s) Na2CO3(s) + 10H2O(g) Statements made by four students about the reaction are given.P. Anhydrous sodium

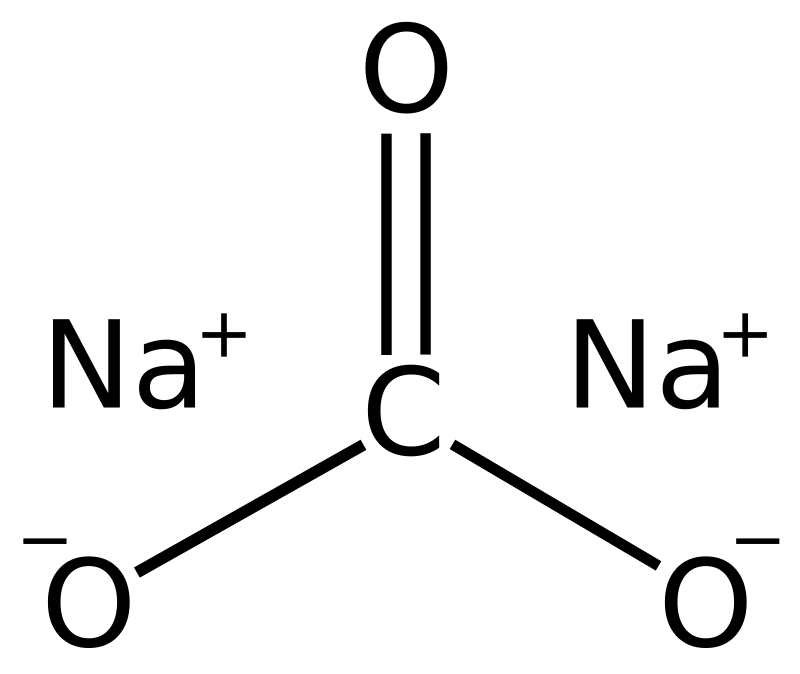
sodium carbonate(Naco3.10H2o)is an important chemical calculate its formula mass in atomic mass unit - Brainly.in

SOLVED: What is the percentage of water in the following compound? Sodium carbonate decahydrate, Na2CO3 • 10H2O % by mass H2O

SOLVED: What is the percentage of water in the following compound? Sodium carbonate decahydrate, Na2CO3 • 10H2O % by mass H2O

Write the chemical name of Na2 CO3.10H2O and Na2CO3, Write the significance of 10H2OMention the term - Brainly.in

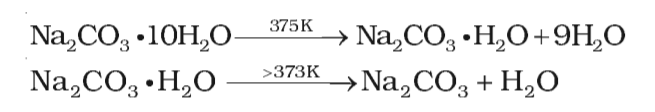
Preparation and thermal properties of Na2CO3·10H2O-Na2HPO4·12H2O eutectic hydrate salt as a novel phase change material for energy storage - ScienceDirect

3.12 gm of washing soda crystal (Na2co3.10h2o) were dissolved in 200 mml of water.20 ml of the - Brainly.in