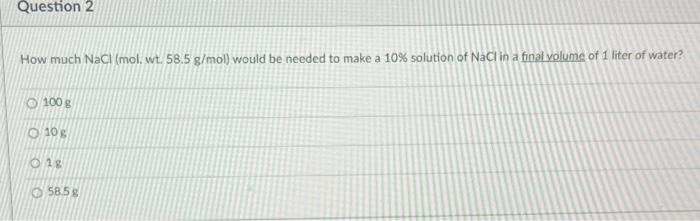
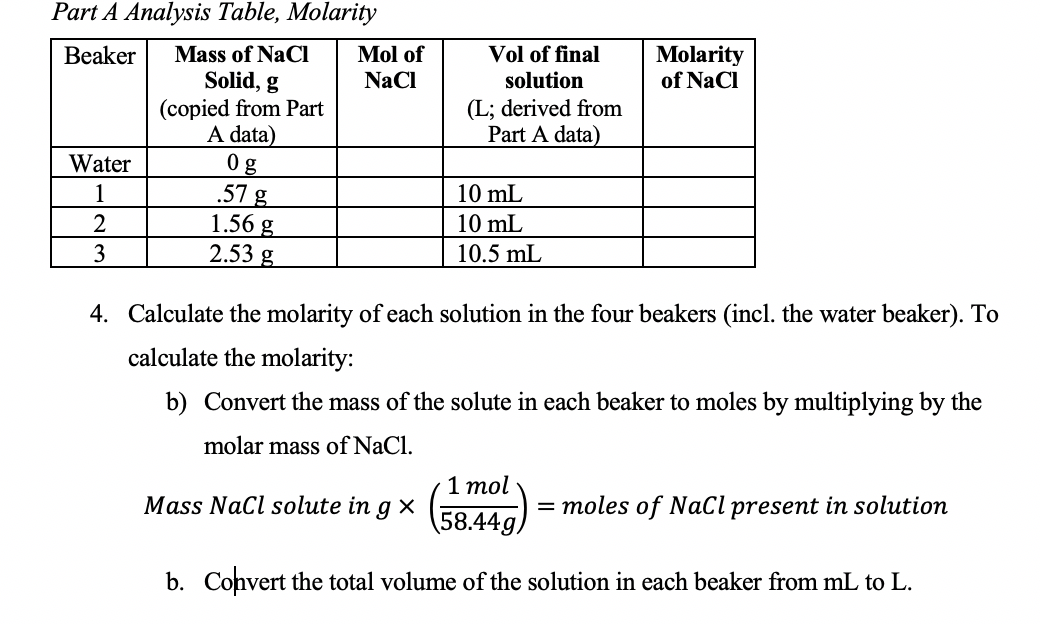
Concentration Calculations Molarity. Objectives To calculate the molecular weight and moles of a substance To calculate the Molarity of a substance using. - ppt download

Formula mass of NaCl is 58.45 g mol ^-1 and density of its pure form is 2.167 g cm ^-3 . The average distance between adjacent sodium and chloride ions in the

Effect of different concentrations of salt (nacl, mol l -1 ) on shoot... | Download Scientific Diagram
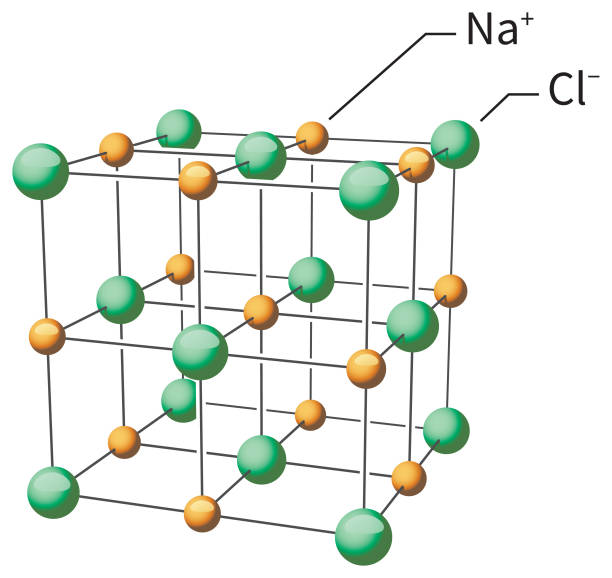
Sodium Chloride Nacl Molecular Structure Stock Illustration - Download Image Now - Grid Pattern, Science, Crystal - iStock

How muCHM molecular mass of `NaCl` is obtained experimentally using colligative properties ? - YouTube