Gypsum (CaSO4.2H2O) mineral, crystal structure. Used for the production of gypsum board, plaster and in fertilizer compositions Stock Photo - Alamy

Calcium sulfate structure. (a) In gypsum (CaSO 4 ·2H 2 O), the water... | Download Scientific Diagram
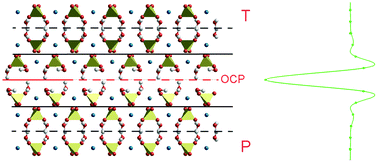
Interfaces structure and stress of gypsum (CaSO4·2H2O) penetration twins - CrystEngComm (RSC Publishing)

Reaction Characteristics of CaSO4/CaSO4·1/2H2O Reversible Reaction for Chemical Heat Pump | Semantic Scholar

Gypsum (CaSO4.2H2O) Mineral, Crystal Structure. Gypsum Is Used For The Production Of Gypsum Board, Plaster And In Fertilizer Compositions. Stock Photo, Picture And Royalty Free Image. Image 17236747.
![Amazon.com: Calcium Sulfate Dihydrate [CaSO4.2H2O] 97% AR Grade Powder 8 Oz in a Space-Saver Bottle USA : Health & Household Amazon.com: Calcium Sulfate Dihydrate [CaSO4.2H2O] 97% AR Grade Powder 8 Oz in a Space-Saver Bottle USA : Health & Household](https://m.media-amazon.com/images/I/818q0xPiqZL._AC_UF1000,1000_QL80_.jpg)
Amazon.com: Calcium Sulfate Dihydrate [CaSO4.2H2O] 97% AR Grade Powder 8 Oz in a Space-Saver Bottle USA : Health & Household

Ethanoic acid on heating with ammonia forms compound A which on treatment with bromine and sodium hydroxide gives compound B . Compound B on treatment with NaNO2/dil.HCl gives compound C . The