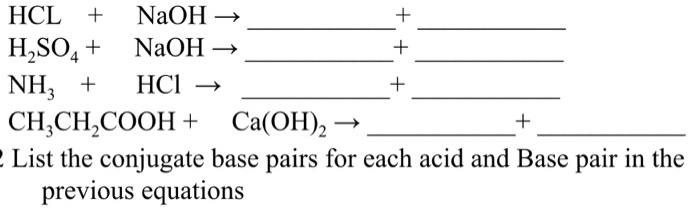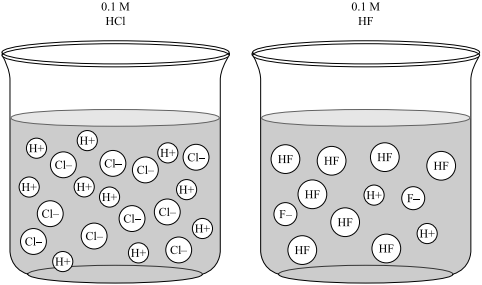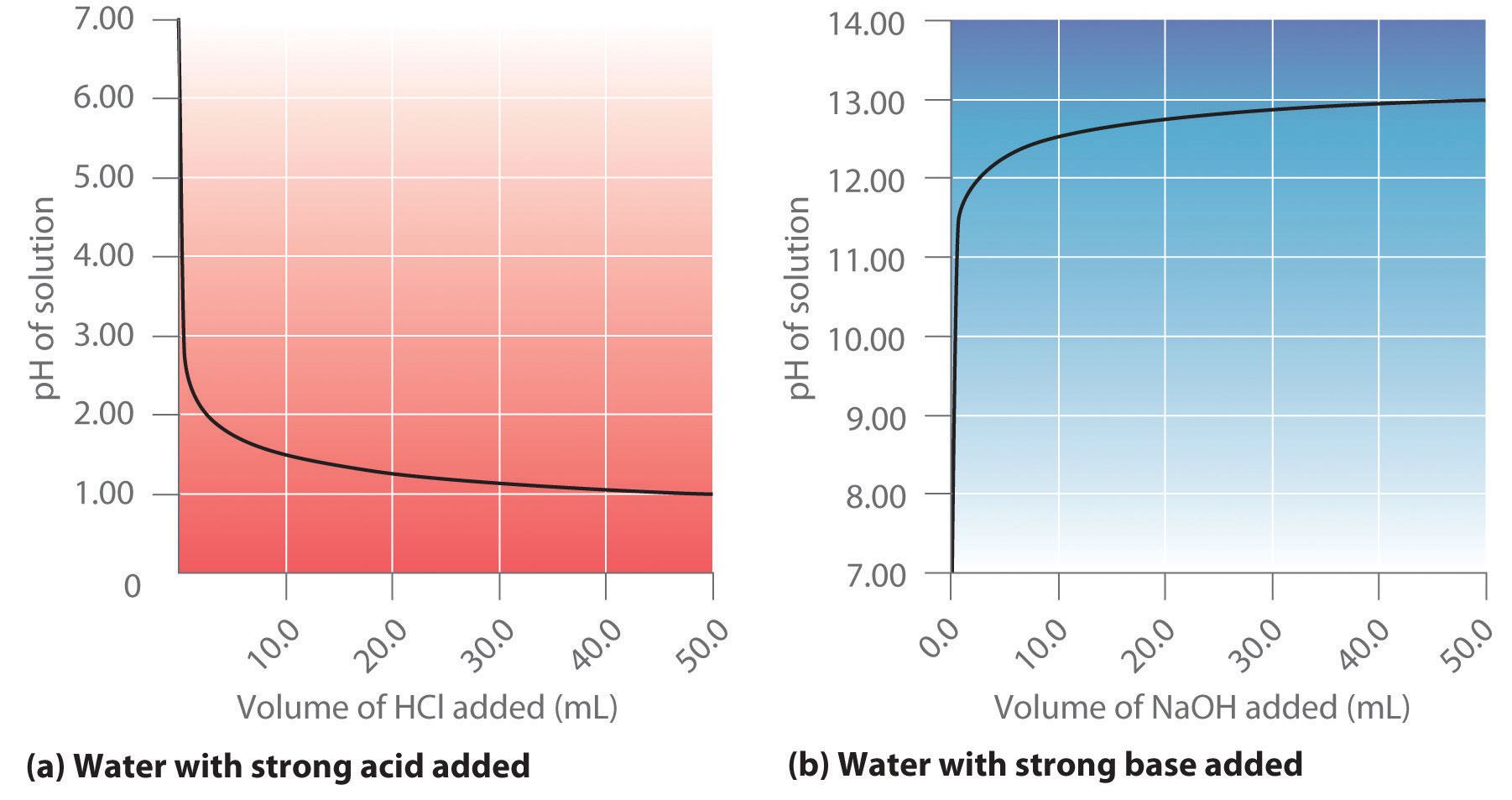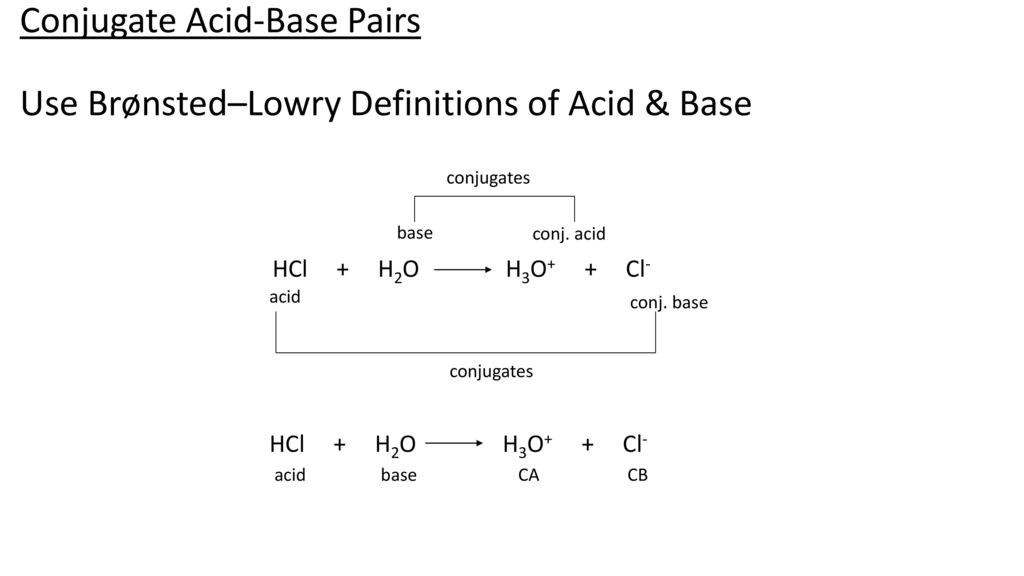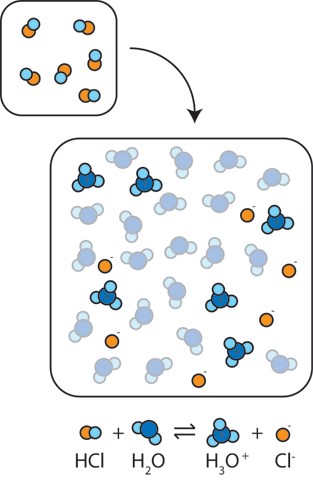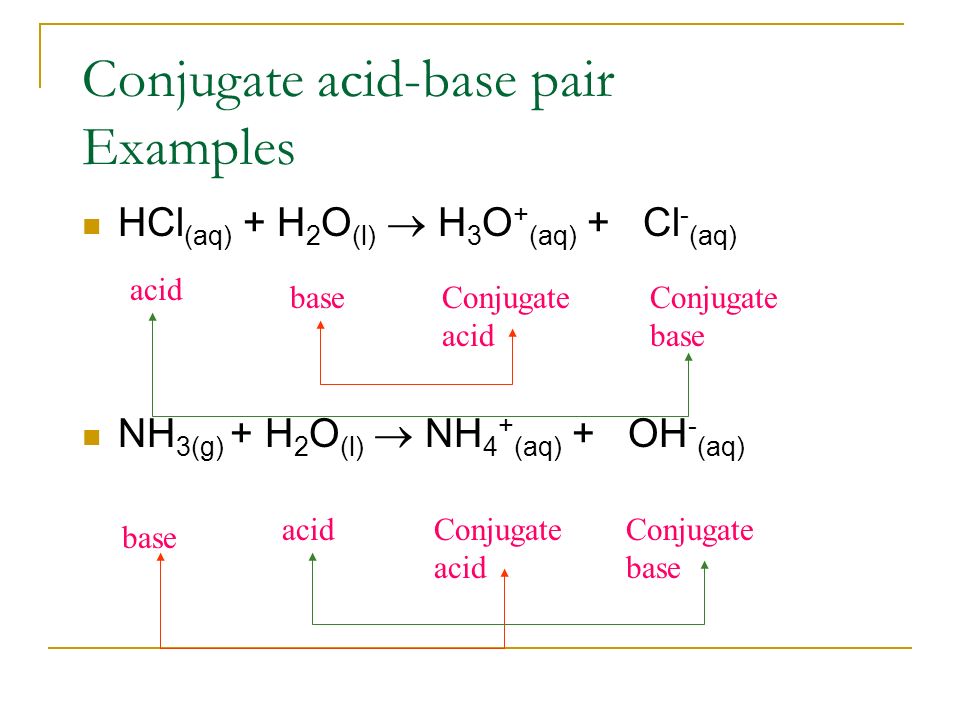
pH and Buffers Acids and Bases Acids: H + donors HCl H + + Cl - CH 3 COOH CH 3 COO - + H + Bases: H + acceptors NaOH + H + Na + + H 2 O - ppt download

Acid – Base Reaction. Chemical Reaction Neutralization The Acid And Base Properties, Producing A Salt And Water.

Write a chemical equation for the acid-base reaction that occurs when p-phenetidine is dissolved in hcl. - Brainly.com